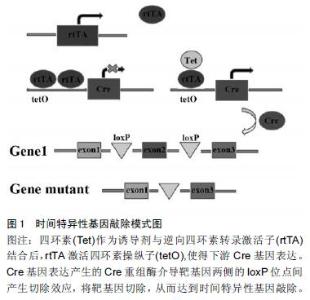
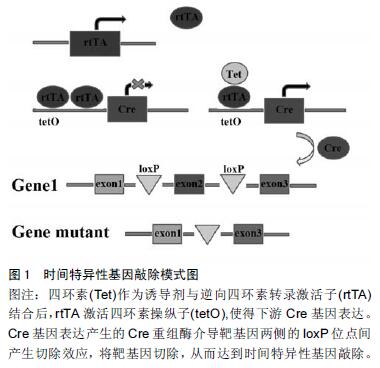
Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (18): 2725-2730.doi: 10.3969/j.issn.2095-4344.2016.18.021
Previous Articles Next Articles
Time-specific gene knockout technology: model establishment, present and future
Chen Rui-jun, Huang Xiao-feng, Zhang Fang-ming
- Department of Stomatology, Beijing Friendship Hospital, Capital Medical University, Beijing 100050, China
-
Received:2016-02-16Online:2016-04-29Published:2016-04-29 -
Contact:Huang Xiao-feng, D.D.S., Ph.D., Professor, Chief physician, Department of Stomatology, Beijing Friendship Hospital, Capital Medical University, Beijing 100050, China -
About author:Chen Rui-jun, Studying for master’s degree, Department of Stomatology, Beijing Friendship Hospital, Capital Medical University, Beijing 100050, China -
Supported by:the National Natural Science Foundation of China, No. 81170943; the Natural Science Foundation of Beijing City, No. 7162053
Cite this article
Chen Rui-jun, Huang Xiao-feng, Zhang Fang-ming. Time-specific gene knockout technology: model establishment, present and future [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(18): 2725-2730.
share this article
| [1] Capecchi MR. Altering the genome by homologous recombination. Science. 1989;244(4910):1288-1292. [2] Capecchi MR. The new mouse genetics: altering the genome by gene targeting. Trends Genet. 1989;5(3): 70-76. [3] Babinet C, Cohen-Tannoudji M. Genome engineering via homologous recombination in mouse embryonic stem (ES) cells: an amazingly versatile tool for the study of mammalian biology. An Acad Bras Cienc. 2001 ;73(3):365-383. [4] Mansour SL. Gene targeting in murine embryonic stem cells: introduction of specific alterations into the mammalian genome. Genet Anal Tech Appl. 1990; 7(8): 219-227. [5] Melton DW. The use of gene targeting to develop animal models for human genetic diseases. Biochem Soc Trans. 1990;18(6):1035-1039. [6] Zimmer A. Manipulating the genome by homologous recombination in embryonic stem cells. Annu Rev Neurosci. 1992;15:115-137. [7] Capecchi MR. Targeted gene replacement. Sci Am. 1994;270(3):52-59. [8] Bradley A, Ramírez-Solis R, Zheng H, et al. Genetic manipulation of the mouse via gene targeting in embryonic stem cells. Ciba Found Symp. 1992;165: 256-269; discussion 269-276. [9] Smith GR. How homologous recombination is initiated: unexpected evidence for single-strand nicks from v(d)j site-specific recombination. Cell. 2004;117(2):146-148. [10] Morrow B, Kucherlapati R. Gene targeting in mammalian cells by homologous recombination. Curr Opin Biotechnol. 1993;4(5):577-582. [11] Waldman AS. Targeted homologous recombination in mammalian cells. Crit Rev Oncol Hematol. 1992;12(1): 49-64. [12] Sollars PJ, Weiser MJ, Kudwa AE, et al. Altered entrainment to the day/night cycle attenuates the daily rise in circulating corticosterone in the mouse. PLoS One. 2014;9(11):e111944. [13] Girardet C, Becquet D, Blanchard MP, et al. Neuroglial and synaptic rearrangements associated with photic entrainment of the circadian clock in the suprachiasmatic nucleus. Eur J Neurosci. 2010; 32(12): 2133-2142. [14] Harmar AJ. An essential role for peptidergic signalling in the control of circadian rhythms in the suprachiasmatic nuclei. J Neuroendocrinol. 2003; 15(4): 335-338. [15] Parkitna JR, Engblom D, Schütz G. Generation of Cre recombinase-expressing transgenic mice using bacterial artificial chromosomes. Methods Mol Biol. 2009;530:325-342. [16] Nagy A, Mar L, Watts G. Creation and use of a cre recombinase transgenic database. Methods Mol Biol. 2009;530:365-378. [17] Wouterlood FG, Bloem B, Mansvelder HD, et al. A fourth generation of neuroanatomical tracing techniques: exploiting the offspring of genetic engineering. J Neurosci Methods. 2014;235:331-348. [18] Król JE, Wojtowicz AJ, Rogers LM, et al. Invasion of E. coli biofilms by antibiotic resistance plasmids. Plasmid. 2013;70(1):110-119. [19] Peng H, Wu Y, Zhang Y. Efficient delivery of DNA and morpholinos into mouse preimplantation embryos by electroporation. PLoS One. 2012;7(8):e43748. [20] Guo L, Ge J, Wang S, et al. A novel method for efficient delivery of stem cells to the ischemic brain. Stem Cell Res Ther. 2013;4(5):116. [21] Krivokharchenko A, Karmenyan A, Sarkisov O, et al. Laser fusion of mouse embryonic cells and intra-embryonic fusion of blastomeres without affecting the embryo integrity. PLoS One. 2012;7(12):e50029. [22] Thomas KR, Capecchi MR. Site-directed mutagenesis by gene targeting in mouse embryo-derived stem cells. Cell. 1987;51(3):503-512. [23] Tsien JZ, Chen DF, Gerber D, et al. Subregion- and cell type-restricted gene knockout in mouse brain. Cell. 1996; 87(7):1317-1326. [24] Sauer B. Inducible gene targeting in mice using the Cre/lox system. Methods. 1998;14(4):381-392. [25] Sauer B. Cre/lox: one more step in the taming of the genome. Endocrine. 2002;19(3):221-228. [26] Gavériaux-Ruff C, Kieffer BL. Conditional gene targeting in the mouse nervous system: Insights into brain function and diseases. Pharmacol Ther. 2007;113(3):619-634. [27] Branda CS, Dymecki SM. Talking about a revolution: The impact of site-specific recombinases on genetic analyses in mice. Dev Cell. 2004;6(1):7-28. [28] Misra RP, Duncan SA. Gene targeting in the mouse: advances in introduction of transgenes into the genome by homologous recombination. Endocrine. 2002;19(3):229-238. [29] Ray MK, Fagan SP, Brunicardi FC. The Cre-loxP system: a versatile tool for targeting genes in a cell- and stage-specific manner. Cell Transplant. 2000; 9(6):805-815. [30] Yoshimura I, Suzuki S, Hayakawa M. Application of Cre-loxP system to the urinary tract and cancer gene therapy. Mol Urol. 2001;5(2):81-84. [31] Rosenberg MP. Gene knockout and transgenic technologies in risk assessment: the next generation. Mol Carcinog. 1997;20(3):262-274. [32] Gu H, Marth JD, Orban PC, et al. Deletion of a DNA polymerase beta gene segment in T cells using cell type-specific gene targeting. Science. 1994;265 (5168):103-106. [33] Sparks MA, Makhanova NA, Griffiths RC, et al. Thromboxane receptors in smooth muscle promote hypertension, vascular remodeling, and sudden death. Hypertension. 2013;61(1): 166-173. [34] Polykratis A, van Loo G, Xanthoulea S, et al. Conditional targeting of tumor necrosis factor receptor-associated factor 6 reveals opposing functions of Toll-like receptor signaling in endothelial and myeloid cells in a mouse model of atherosclerosis. Circulation. 2012;126(14):1739-1751. [35] Wei Y, Nazari-Jahantigh M, Neth P, et al. MicroRNA-126, -145, and -155: a therapeutic triad in atherosclerosis? Arterioscler Thromb Vasc Biol. 2013; 33(3):449-454. [36] Lin CI, Barletta JA, Nehs MA, et al. Thyroid-specific knockout of the tumor suppressor mitogen-inducible gene 6 activates epidermal growth factor receptor signaling pathways and suppresses nuclear factor-κB activity. Surgery. 2011;150(6):1295-1302. [37] Morioka Y, Isotani A, Oshima RG, et al. Placenta- specific gene activation and inactivation using integrase-defective lentiviral vectors with the Cre/LoxP system. Genesis. 2009;47(12):793-798. [38] Banasik MB, McCray PB Jr. Integrase-defective lentiviral vectors: progress and applications. Gene Ther. 2010;17(2):150-157. [39] Staunstrup NH, Mikkelsen JG. Integrase-defective lentiviral vectors--a stage for nonviral integration machineries. Curr Gene Ther. 2011;11(5):350-362. [40] Negri DR, Michelini Z, Bona R, et al. Integrase-defective lentiviral-vector-based vaccine: a new vector for induction of T cell immunity. Expert Opin Biol Ther. 2011;11(6):739-750. [41] Liu KC, Lin BS, Gao AD, et al. Integrase-deficient lentivirus: opportunities and challenges for human gene therapy. Curr Gene Ther. 2014;14(5):352-364. [42] Ward JM, Elmore SA, Foley JF. Pathology methods for the evaluation of embryonic and perinatal developmental defects and lethality in genetically engineered mice. Vet Pathol. 2012;49(1):71-84. [43] Chu L, Li J, Liu Y, et al. Targeted gene disruption in zebrafish reveals noncanonical functions of LH signaling in reproduction. Mol Endocrinol. 2014;28(11):1785-1795. [44] Kumar TR. What have we learned about gonadotropin function from gonadotropin subunit and receptor knockout mice? Reproduction. 2005;130(3):293-302. [45] Shimizu E, Tang YP, Rampon C, et al. NMDA receptor-dependent synaptic reinforcement as a crucial process for memory consolidation. Science. 2000;290(5494):1170-1174. [46] Werfel S, Jungmann A, Lehmann L, et al. Rapid and highly efficient inducible cardiac gene knockout in adult mice using AAV-mediated expression of Cre recombinase. Cardiovasc Res. 2014;104(1):15-23. [47] Sabharwal R, Chapleau MW. Autonomic, locomotor and cardiac abnormalities in a mouse model of muscular dystrophy: targeting the renin-angiotensin system. Exp Physiol. 2014;99(4):627-631. [48] Heine HL, Leong HS, Rossi FM, et al. Strategies of conditional gene expression in myocardium: an overview. Methods Mol Med. 2005;112:109-154. [49] Choi CI, Yoon SP, Choi JM, et al. Simultaneous deletion of floxed genes mediated by CaMKIIα-Cre in the brain and in male germ cells: application to conditional and conventional disruption of Goα. Exp Mol Med. 2014;46:e93. [50] Mo L, Cheng J, Lee EY, et al. Gene deletion in urothelium by specific expression of Cre recombinase. Am J Physiol Renal Physiol. 2005;289(3):F562-568. [51] Casanova E, Fehsenfeld S, Mantamadiotis T, et al. A CamKIIalpha iCre BAC allows brain-specific gene inactivation. Genesis. 2001;31(1):37-42. [52] Bex A, Vooijs M, Horenblas S, et al. Controlling gene expression in the urothelium using transgenic mice with inducible bladder specific Cre-lox recombination. J Urol. 2002;168(6):2641-2644. [53] Castrop H, Oppermann M, Weiss Y, et al. Reporter gene recombination in juxtaglomerular granular and collecting duct cells by human renin promoter-Cre recombinase transgene. Physiol Genomics. 2006; 25(2):277-285. [54] Kistner A, Gossen M, Zimmermann F, et al. Doxycycline-mediated quantitative and tissue-specific control of gene expression in transgenic mice. Proc Natl Acad Sci U S A. 1996;93(20):10933-10938. [55] Perl AK, Wert SE, Nagy A, et al. Early restriction of peripheral and proximal cell lineages during formation of the lung. Proc Natl Acad Sci U S A. 2002;99(16): 10482-10487. [56] Xie W, Chow LT, Paterson AJ, et al. Conditional expression of the ErbB2 oncogene elicits reversible hyperplasia in stratified epithelia and up-regulation of TGFalpha expression in transgenic mice. Oncogene. 1999;18(24):3593-3607. [57] Goldfless SJ, Belmont BJ, de Paz AM, et al. Direct and specific chemical control of eukaryotic translation with a synthetic RNA-protein interaction. Nucleic Acids Res. 2012;40(9):e64. [58] Meitert J, Aram R, Wiesemann K, et al. Monitoring the expression level of coding and non-coding RNAs using a TetR inducing aptamer tag. Bioorg Med Chem. 2013; 21(20):6233-6238. [59] Belmont BJ, Niles JC. Inducible control of subcellular RNA localization using a synthetic protein-RNA aptamer interaction. PLoS One. 2012;7(10):e46868. [60] Chaiyachati BH, Kaundal RK, Zhao J, et al. LoxP-FRT Trap (LOFT): a simple and flexible system for conventional and reversible gene targeting. BMC Biol. 2012;10:96. |
| [1] | Qu Yuan-qian, Zhou Qing, Zhang Jin, Xu San-rong. Inhibiting colorectal cancer liver metastasis by combined injection of allogeneic bone marrow cells and lymphocytes [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(25): 4053-4058. |
| [2] | Luo Su-yi, Huang Wei, Wang Jing, Wang Xi-yun, Li Jin-tao. Effects of tropomyosin 4 applied in spinal cord injuries via lentiviral vector recombination and the underlying mechanism: study protocol for a randomized controlled trial [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(37): 5573-5579. |
| [3] | Wei Wei, Li Xiao-ling, Yuan Wen-chang. Construction and application of Staphylococcus aureus gene knockout plasmid [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(5): 799-804. |
| [4] | Li Lin-ge, Feng Juan, Hu Bin, Shou Xi, Zhang Chun, Tian Yu, Jiang Chun-rong, Zhang Yu, Zhang Hua . Establishing mouse models of allergic rhinitis by knocking out H2-eb1 gene [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(27): 4417-4422. |
| [5] | Liu Guang-rong, Chen Qing-yuan, Hu Guan-song, Cai Yu-chun. Surface markers and skin differentiation of epidermal stem cells: theoretical features and clinical feasibility [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(28): 4573-4577. |
| [6] | Du Qi-cui, Xiao Wen-lin, Sun Gui-lan, Ma Xiu-xing, Yao Ru-yong. Primary culture of human hypertrophic scar fibroblasts and its biological behavior [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(7): 1174-1179. |
| [7] | He Bao-li, Ma Li-hua, Chen Li-ling, Liu Ru-wen, Yang Ren-hua. Establishment of human-rhesus chimeric liver using adult bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(45): 7827-7833. |
| [8] | Mori Gele, Shao Zeng-wu. Restraining degeneration of intervertebral discs by transplantation of Notch-1knockout rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(36): 6403-6408. |
| [9] | Zhang Xian-yu, Wang Wei, Chen Wei, Chen Jian-mei, Xu Hao . Autologous bone marrow mesenchymal stem cells and related chemokines in fracture microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(27): 5070-5079. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||